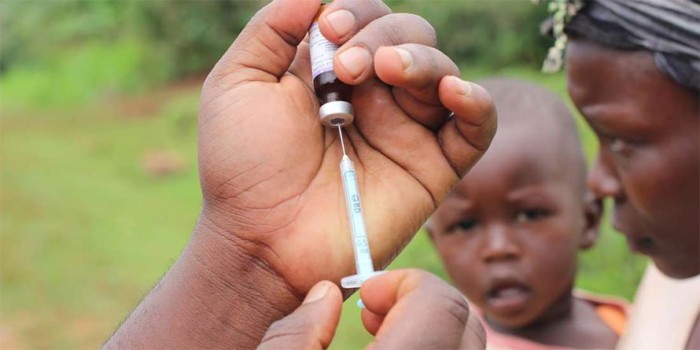
The government has denied being aware of any bid to have the COVID-19 vaccine tested in Kenya.
In a press briefing in Nairobi, Health CAS Rashid Aman said that they are not privy to such a development in the country.
He pointed out that if there is such an initiative there are procedural guidelines that ought to be followed contrary to what is being alleged.
“We do not have any communication on this at the moment but if and when we do, there are certain, laid out procedures which must be followed,” Aman said.
“Should the team from Oxford reach out to the Kenyan government, then there is a very robust process to be followed by the pharmacy and poisons board before the vaccine can be approved for a clinical trial,” He added.
The matter has been brimming in social media in the country for the better part of Friday after An article published by the BBC on Thursday, revealed that the UK is considering testing the vaccine in Kenya.
“The Oxford team is also considering a vaccine trial in Africa, possibly in Kenya, where the rates of transmission are growing from a lower base.”
In an interview with the BBC, the media house’s Medical Correspondent Fergus Walsh also revealed that the UK is considering a try out of the vaccine in the country.
“The team in Oxford has a solid record going back 30 years, and they have developed a lot of successful prototype vaccines against another type of Coronavirus, MARS which has done well in clinical trials.
“They have also developed vaccines against the plague and malaria. If they don’t get early quick results in the UK, they are considering a trial in Kenya where the epidemic of Coronavirus may well be on the rise.
“This virus is known, this vaccine is known to produce a strong antibody response but that doesn’t necessarily equate to protection, we are going to need many vaccines, there are dozens in development,” said Walsh.
scientists from Oxford commenced its first human trials on Thursday, April 23 with two volunteers injected.
The two who are scientists were the first batch of more than 800 people recruited for the study where half will receive the Covid-19 vaccine and half a control vaccine which protects against meningitis but not coronavirus.7
The volunteers will not know which of the two vaccines they are getting but the doctors conducting the trial will.